How to talk like a chemist: Hydroxide, hydroxyl? Aren't they the same thing?
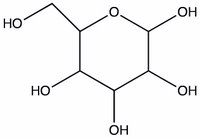 The glucose molecule shown at left has lots of OH's on it. Are these hydroxides? Technically, no. Hydroxides are ions, individual OH- units, either in solution or in a crystal. In glucose, the OHs are termed hydroxyl groups, where the OH is bound covalently to the molecule. The name gives a clue to the chemical structure and expected reactivity!
The glucose molecule shown at left has lots of OH's on it. Are these hydroxides? Technically, no. Hydroxides are ions, individual OH- units, either in solution or in a crystal. In glucose, the OHs are termed hydroxyl groups, where the OH is bound covalently to the molecule. The name gives a clue to the chemical structure and expected reactivity!Alchohols have as a distinguishing feature a hydroxyl group, and therefore don't behave as bases (losing hydroxide ions) like sodium hydroxide would, even though their chemical formulas might look similar. For example, compare the formulas for sodium hydroxide (NaOH) and ethyl alchohol (CH3CH2OH).


1 Comments:
This is very clear! I appreciate it! It's really helpful.
Post a Comment
<< Home